Difference Urine vs Oral Tests: Best Choice Explained

Finding a reliable and affordable drug testing method is a challenge every North American clinic and staffing agency faces amid rising compliance demands. Your choice directly affects program efficiency, result accuracy, and cost control. Urine tests typically detect drug use over several days, while oral fluid testing captures more recent use with shorter windows and stronger tamper resistance. This guide will help you compare detection windows and accuracy factors so you can match the right test to your operational and regulatory needs. In this article, we’ll specifically look at the comparison between Urine vs Oral Tests.
Understanding the Differences Between Urine vs Oral Tests
Table of Contents
- Detection Windows and Key Accuracy Factors
- How Sample Collection and Adulteration Differ
- Regulatory Guidelines and Legal Considerations
- Cost, Use Cases, and Choosing the Right Test
Key Takeaways
| Point | Details |
|---|---|
| Detection Windows Impact Choices | Selecting the appropriate testing method affects the detection window of drug activity, with urine tests offering longer historical insights compared to oral tests. |
| Accuracy Factors | Test accuracy is influenced by biological factors, specimen type, and analytical methods, necessitating careful selection based on organizational needs. |
| Regulatory Compliance | Adhering to federal and state regulations is crucial when employing drug tests, as they dictate acceptable methods and documentation practices. |
| Cost vs. Use Case Alignment | Evaluating costs alongside specific use cases ensures optimized testing strategies, balancing budget constraints with the need for effective detection methods. |
…
Detection Windows and Key Accuracy Factors
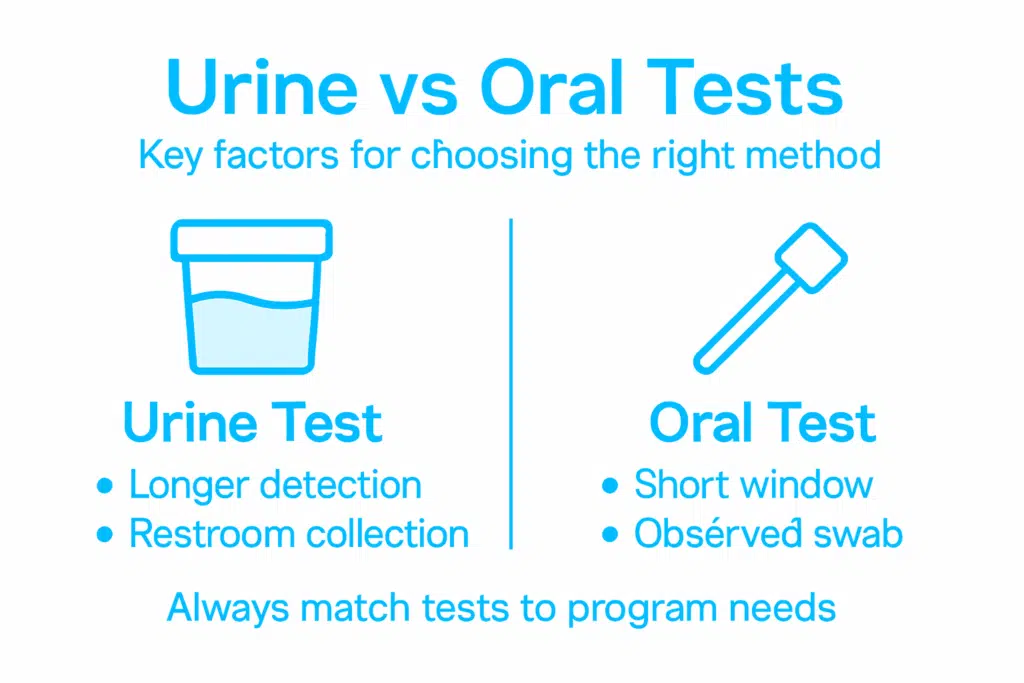
When you’re selecting a drug testing method for your clinic or staffing agency, understanding detection windows becomes critical to your testing strategy. The timeframe during which a test can reliably detect drug use varies dramatically between urine and oral testing methods. Urine tests excel at capturing a broader historical window because metabolites, the byproducts of drug metabolism, remain detectable in the body for extended periods. Oral fluid tests, by contrast, detect parent drugs and recent metabolites with shorter detection periods, typically capturing drug use within hours to approximately one day. This fundamental difference means your choice of testing method directly impacts what window of drug activity you’re actually measuring.
Several biological and practical factors influence how accurately each test performs within its detection window. Detection windows depend on drug type, frequency of use, individual metabolism, and specimen type. For urine testing specifically, hydration levels, metabolic rates, urinary pH, and the particular drug’s chemical properties all play significant roles in accuracy. A dehydrated employee may show a more concentrated sample, potentially affecting results. Metabolism varies widely among individuals due to age, body weight, liver function, and genetic factors. These variables mean two people using the same substance may have completely different detection timelines. Oral fluid tests are less influenced by these metabolic variations, offering more consistent detection windows across different individuals, though with shorter overall detection periods.
Accuracy also depends on the analytical methods and thresholds your testing supplies use. Modern immunoassay cups and dip cards from reputable manufacturers offer screening reliability, but confirmation testing through more sophisticated methods provides definitive results. When you’re procuring bulk testing supplies, understanding these nuances helps you select the right panel counts and testing formats for your program’s objectives. Some programs need to detect recent use only, making oral tests ideal. Others require broader historical screening, where urine testing proves more effective. Consider your program’s timeline, staff turnover, and regulatory requirements when evaluating detection window capabilities.
Pro tip: Build your procurement strategy around your actual testing window needs rather than defaulting to one method, and document your detection window expectations with clients and employees so results are interpreted correctly.
To clarify key differences, here’s a side-by-side comparison of urine and oral drug testing methods:
| Aspect | Urine Testing | Oral Fluid Testing |
|---|---|---|
| Detection Window | 1-4 days for most drugs | Few hours to 24 hours |
| Susceptibility to Tampering | Higher; many external adulteration methods | Lower; direct observation reduces risk |
| Collection Setting | Requires restroom; often unobserved | Simple; directly observed collection |
| Donor Comfort | May feel invasive to some | Generally more comfortable |
How Sample Collection and Adulteration Differ
The collection process itself creates a fundamental operational difference between urine and oral drug tests. Urine collection typically occurs in a bathroom or private area with minimal observation, which creates both logistical ease and vulnerability to tampering. Your staff collects the sample with limited direct supervision, meaning the specimen can be exposed to various forms of interference. Oral fluid collection, conversely, happens in a controlled observed setting where the donor provides the sample under direct watch. This observed nature dramatically reduces the opportunity for sample substitution or external contamination. For procurement managers running high-volume testing programs, this distinction impacts your collection protocols, staff training requirements, and facility design. Observed collection for oral tests typically requires less specialized bathroom infrastructure compared to urine collection, where you need monitored restrooms to prevent adulteration attempts.

Adulteration methods differ significantly between these two specimen types, which means your testing supplies must address different integrity threats. Urine samples can be compromised by internal dilution through excessive fluid intake, external adulteration by adding chemicals like bleach or nitrites, or substitution with synthetic urine or another person’s sample. Detecting these tampering methods requires specimen validity testing that checks for abnormal creatinine levels, pH variations, oxidants, and specific gravity. Oral fluid samples face lower adulteration risk because of the observed collection process, though external contamination remains theoretically possible. Your testing supplies should include adulterant detection capabilities appropriate to each specimen type. Urine test cups with integrated validity markers for creatinine, oxidants, and pH provide screening protection against the most common tampering attempts.
Understanding these collection and adulteration differences helps you select the right testing format for your organization’s risk tolerance and compliance needs. Urine testing requires robust specimen handling protocols and staff training on collection security to combat the higher adulteration risk. Oral testing’s observed collection reduces these concerns but may feel less comfortable for some donors depending on your clientele and testing context. When procuring bulk supplies, consider your facility’s ability to conduct observed collections, your tolerance for potential specimen tampering, and regulatory requirements specific to your industry. Some programs need maximum security against adulteration, making urine testing with validity markers essential. Others prioritize donor comfort and faster results, where oral testing’s lower contamination risk and simpler collection become attractive advantages.
Pro tip: Stock both urine test cups with integrated validity markers and oral swab kits, then match the testing method to specific program needs rather than relying on a single approach for all donor populations.
Regulatory Guidelines and Legal Considerations
Navigating the regulatory landscape for drug testing is non-negotiable when you’re procuring supplies for clinics or staffing agencies. Federal regulations shape what testing methods you can use, how you collect samples, and what documentation you must maintain. The Department of Transportation established the foundation for federally regulated workplace testing through 49 CFR Part 40, which has historically focused on urine testing. However, the landscape is evolving. DOT regulations now authorize oral fluid testing as an alternative to urine drug testing in federally regulated workplaces, though implementation continues as laboratories complete certification requirements. This authorization means your organization can now select oral testing for certain federally covered positions, provided you use certified laboratories and maintain strict collection and chain-of-custody protocols.
Beyond DOT regulations, the Substance Abuse and Mental Health Services Administration (SAMHSA) establishes Mandatory Guidelines for federal workplace drug testing that apply broadly across government agencies and contractors. These guidelines specify acceptable testing panels, medical review officer processes, specimen collection procedures, chain-of-custody requirements, and confidentiality protections. Whether you choose urine or oral testing, your collection procedures and staff training must comply with these established standards. The guidelines ensure consistency and fairness while protecting employee rights. Your procurement strategy must account for these compliance requirements when selecting testing supplies. Urine test cups and oral swab kits must be from manufacturers who understand and comply with FDA approval and CLIA waiver requirements for forensic and clinical use. When you source supplies from reputable distributors, you gain confidence that your testing infrastructure meets regulatory expectations.
State and local regulations may impose additional requirements beyond federal standards, making compliance more complex. Some states have stricter guidelines on testing procedures, result reporting timelines, or employee notification requirements. Healthcare facilities operate under different regulatory frameworks than staffing agencies, requiring different testing supply specifications. Your legal team should review applicable regulations before finalizing your testing program design and supply procurement decisions. Consider consulting with medical review officers and compliance specialists who understand your specific industry sector and geographic footprint. The regulatory environment continues evolving, particularly around oral testing authorization and laboratory certification requirements. Building flexibility into your testing program by stocking multiple testing methods positions you to adapt as regulations change without disrupting operations.
Pro tip: Document your regulatory compliance framework in writing and review it annually with legal counsel, ensuring your procured testing supplies maintain proper FDA approvals and CLIA waivers for your specific use case.
Cost, Use Cases, and Choosing the Right Test
When you’re building a drug testing program, cost directly impacts your decision between urine and oral testing methods. Urine testing remains the most economical option at scale, particularly when you’re purchasing bulk supplies. Immunoassay screening methods for urine deliver rapid point-of-care results at minimal per-test cost, making them ideal for high-volume programs operating under tight budget constraints. Oral fluid testing carries higher upfront costs due to equipment requirements and the current limitations in laboratory certification across the country. However, cost cannot be your only consideration. Your use case, donor population, and testing objectives must align with the method you choose. Urine drug tests are favored for their broad availability and relatively low cost, making them practical for long-term monitoring programs like substance use disorder treatment or workplace compliance screening. Oral testing shines in scenarios where you need rapid detection of recent use, such as impairment assessment or post-incident testing, where the higher per-test cost justifies the immediacy and donor comfort benefits.
Your specific use case determines which test method delivers the most value. Clinical settings monitoring patients on long-term opioid therapy or substance use disorder treatment typically favor urine testing because it provides historical insight over days and supports consistent protocol implementation. Staffing agencies conducting pre-employment screening or random workplace testing benefit from urine testing’s lower cost and established chain-of-custody procedures that satisfy regulatory expectations. Law enforcement and impairment-based testing contexts increasingly use oral testing because it detects recent drug use more reliably and occurs under observed conditions that prevent tampering. Treatment facilities managing patient populations may use both methods strategically, deploying oral testing for intake assessments or suspected recent use and urine testing for ongoing monitoring. The key is matching your program’s objectives to the detection window and operational characteristics that serve your mission best.
Choosing the right test requires evaluating five critical factors together. First, consider your detection window needs: Do you need historical perspective over several days, or recent use detection within hours? Second, assess cost per test including supplies, collection, analysis, and staff time. Third, evaluate your facility infrastructure: Can you conduct observed collections, or do you need minimal supervision options? Fourth, review donor populations and comfort: Will your participants accept oral collection, or does your facility need faster, less intrusive methods? Fifth, confirm regulatory compliance: Does your jurisdiction and industry sector permit the method you’re considering? Most successful programs maintain inventory of both urine and oral testing supplies, deploying each method strategically based on individual circumstances rather than relying exclusively on one approach. This flexibility allows you to serve different program needs without compromising cost efficiency or regulatory compliance.
The following table summarizes which test type aligns best with common testing scenarios:
| Use Case | Best Fit Test Type | Key Reason |
|---|---|---|
| Historical substance monitoring | Urine | Extended detection window |
| Post-incident/impairment assessment | Oral Fluid | Recent use detection |
| Random workplace screening | Urine | Lower cost, regulatory familiarity |
| Intake or entry assessments | Oral Fluid | Tamper-resistant, rapid results |
Pro tip: Calculate your true cost per test by including supplies, staff labor, results processing, and potential retesting, then compare oral and urine methods across your anticipated annual volume to identify which combination optimizes both budget and program effectiveness.
Choose the Right Drug Testing Supplies for Your Program’s Success
Choosing between urine and oral drug testing presents real challenges. The detection windows, risk of adulteration, regulatory compliance, and cost all affect your decision-making. At the core, your goal is clear: secure accurate, reliable, and cost-effective testing kits that fit your unique operational needs and compliance demands. Whether your program requires the broad detection window of urine tests or the tamper-resistant convenience of oral swabs, having trusted suppliers with a wide selection and expert support is essential.
Slash Medical offers over 400 FDA-approved and CLIA-waived products including urine and oral drug test cups, dip cards, and adulterant detection kits designed to meet your program’s exact standards. We provide bulk purchasing, private label manufacturing, and drop shipping solutions tailored for clinics, staffing agencies, treatment centers, and government organizations. With competitive pricing and nationwide shipping, you can confidently stock the right inventory that aligns with your detection window goals and regulatory framework.
Take control of your drug testing program today by exploring our comprehensive catalog. Browse Slash Medical’s inventory now and discover how to optimize your test supply strategy to reduce costs and increase testing accuracy. Visit Slash Medical and equip your organization with the right tools to handle urine and oral testing demands reliably and efficiently.
Find the exact drug test kits your program needs and streamline procurement with Slash Medical’s trusted solutions. Start ordering today for dependable drug testing supplies delivered when and where you need them.
Frequently Asked Questions
What are the main differences in detection windows between urine and oral tests?
Urine tests typically have a detection window of 1-4 days for most drugs, while oral fluid tests can detect drug use within a few hours to 24 hours after use. This means urine tests provide a broader historical perspective, whereas oral tests focus on more recent use.
How does sample collection differ between urine and oral testing methods?
Urine testing is often conducted in a restroom with minimal supervision, which increases the risk of tampering. Conversely, oral fluid testing occurs in a controlled environment with direct observation, significantly reducing the chances of sample contamination or substitution.
What factors can affect the accuracy of urine and oral fluid drug tests?
The accuracy of urine tests can be influenced by factors such as hydration levels, metabolic rates, and urinary pH. Oral fluid tests, however, offer more consistent results across individuals, primarily because they detect recent drug use under closely monitored conditions.
When is it more appropriate to use urine testing instead of oral fluid testing?
Urine testing is preferable when historical drug use detection is essential, such as in long-term monitoring programs or comprehensive workplace compliance screening. It is also more cost-effective for high-volume testing scenarios compared to oral fluid testing.
